Alzheimer’s disease is a relentless neurodegenerative condition that affects millions of people worldwide. Characterized by cognitive decline, memory loss, and a host of other neurological symptoms, it significantly impacts patients, their families, and healthcare systems. A diagnosis of Alzheimer’s disease can feel like a race against time due to the progressive nature of the condition. One of the most common questions neurologists encounter is, “Can you slow the progression of Alzheimer’s disease?” In this blog, we hope to provide some insight into how Alzheimer’s disease affects the brain and what can be done.
Understanding Alzheimer’s Disease
To understand potential interventions, it’s crucial to first grasp what happens in the brain during Alzheimer’s disease. It is characterized by the accumulation of amyloid plaques and tau tangles, which disrupt neural communication and lead to cell death. This damage typically begins in the hippocampus—the brain’s memory center—before spreading to other areas.
Amyloid Plaques
Amyloid plaques are sticky buildups which accumulate outside and around neurons. They are composed primarily of a protein fragment called beta-amyloid (Aβ). This fragment is snipped from a larger protein called amyloid precursor protein (APP), which is involved in neuron growth, repair, and other tasks crucial for normal brain function.
In Alzheimer’s disease, something goes wrong with the cutting process, leading to the production of a version of beta-amyloid that has a tendency to clump together. These clumps begin to gather into plaques that can disrupt cell function in multiple ways:
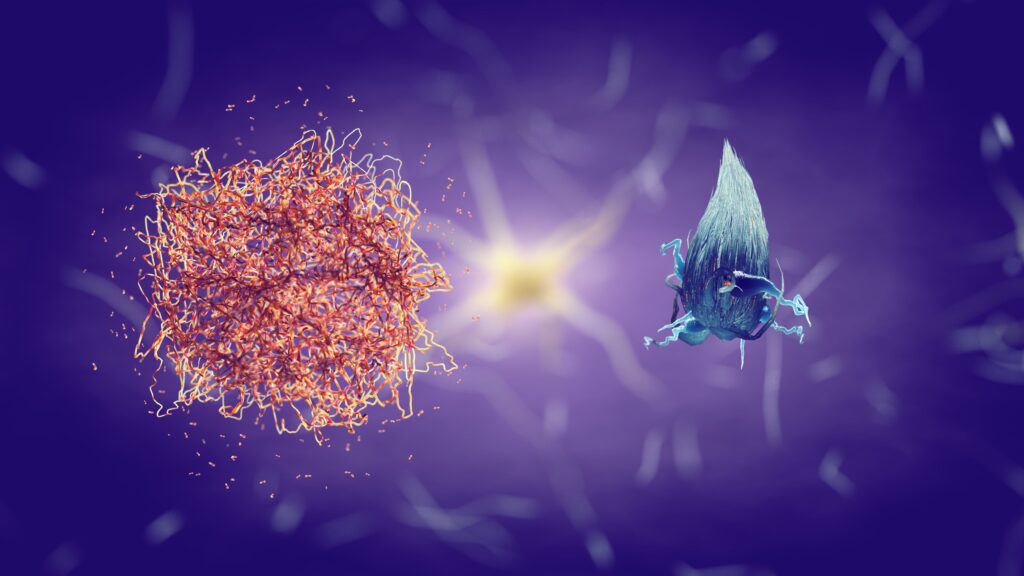
- Interfering with neuron communication
- Triggering immune system responses
- Inducing neuron death
Tau Tangles
Tau tangles are abnormal accumulations of a protein called tau, which occurs inside brain cells. Tau usually functions as a stabilizer for microtubules, which are like the skeleton and transport network for nutrients within the neuron. However, in Alzheimer’s disease, tau proteins undergo abnormal chemical changes, becoming hyperphosphorylated, causing them to detach from microtubules and stick to other tau molecules.
These tangles disrupt the neuron’s transport system, which can cause a cascade of problems:
- Impaired nutrient transport
- Cell death
The relationship between amyloid plaques and tau tangles is complex. Some researchers believe that the formation of amyloid plaques may be an early trigger for the subsequent development of tau tangles, contributing to the progression of Alzheimer’s disease. However, this relationship is not entirely linear or clear-cut, and it is a significant focus of current research.
Current Treatments and Interventions
While there is currently no cure for Alzheimer’s, some treatments can help slow the disease’s progression. These treatments usually aim to improve symptoms or modify the underlying disease processes.
Medications
FDA-approved drugs for Alzheimer’s disease fall into two broad categories: those that aim to modify the disease progression and those that treat the symptoms of the disease. Disease-modifying drugs are designed to change the underlying biology of Alzheimer’s, potentially slowing down the rate at which the disease progresses. These drugs work by targeting the disease mechanisms, such as the buildup of amyloid plaques or tau tangles, that are believed to contribute to Alzheimer’s disease. The efficacy of disease-modifying drugs can be challenging to determine, as their goal is to influence long-term outcomes like cognitive decline and daily functioning. On the other hand, symptomatic drugs do not influence the disease course but instead focus on managing the symptoms experienced by patients.
The United States Food and Drug Administration (FDA) has approved two classes of medications to treat the cognitive symptoms associated with Alzheimer’s disease. The first class includes cholinesterase inhibitors, which are typically prescribed for mild to moderate Alzheimer’s cases. These drugs work by preventing the breakdown of acetylcholine, a neurotransmitter that is important for learning and memory. This class includes donepezil, rivastigmine, and galantamine.
The second class is the NMDA (N-methyl-D-aspartate) receptor antagonist, represented by memantine, which is approved for the treatment of moderate to severe Alzheimer’s disease. Memantine regulates the activity of glutamate, another neurotransmitter that, when produced in excessive amounts, can lead to brain cell death. By moderating the toxic effects associated with high levels of glutamate, memantine helps to slow the progression of symptoms related to Alzheimer’s. Both types of medications aim to manage symptoms and improve the quality of life for individuals with Alzheimer’s but do not cure or reverse the disease.
Diet

Dietary habits have long been studied for their potential impact on the progression of Alzheimer’s disease, with some research suggesting that what we eat may influence brain health. Diets rich in antioxidants, such as the Mediterranean diet, which emphasizes fruits, vegetables, whole grains, nuts, fish, and olive oil, have been associated with a reduced risk of cognitive decline. These foods provide vital nutrients that can help protect the brain against oxidative stress, inflammation, and vascular damage — all conditions that contribute to Alzheimer’s disease. In fact, one study found that people who ate a Mediterranean diet had less of the proteins responsible for Alzheimer’s disease than people who ate an American diet.
Additionally, certain nutrients like omega-3 fatty acids, found in fish, have been shown to support brain cell structure and function. Vitamin E, found in nuts and seeds, is another antioxidant that may help protect neurons. While no diet can prevent or cure Alzheimer’s, a balanced, nutrient-rich eating pattern can potentially support brain health and slow the progression of cognitive decline by mitigating risk factors and supporting overall brain function.
Exercise
Regular physical exercise is increasingly recognized as a potential non-pharmacological intervention to slow the progression of Alzheimer’s disease. Exercise may confer neuroprotective effects through several mechanisms: it enhances blood flow to the brain, which improves neuronal health and function, and it may also elevate levels of brain-derived neurotrophic factor (BDNF), a protein that supports the survival of existing neurons and encourages the growth of new neurons and synapses. Furthermore, exercise stimulates the release of other neurochemicals that are involved in cognitive function, mood regulation, and the reduction of stress hormones. Aerobic activities, in particular, have been shown to reduce brain atrophy and can lead to improvements in memory and executive function, which are areas typically affected by Alzheimer’s. By incorporating regular physical activity into a daily routine, individuals may be able to delay the onset of Alzheimer’s symptoms and slow the rate of cognitive decline.
Cognitive Training
Cognitive training represents a promising avenue for potentially slowing the progression of Alzheimer’s disease by engaging the brain in tasks that can improve its function. This form of mental exercise is aimed at enhancing cognitive reserves, the brain’s ability to improvise and find alternate ways of completing tasks when faced with challenges. Activities such as puzzles, memory games, learning a new language, or playing a musical instrument involve complex thinking and problem-solving skills that can reinforce neural connections and promote new ones. Studies suggest that cognitive training can lead to improved performance in specific cognitive tasks and may also have a generalizable impact on overall mental function. By continually challenging and stimulating the brain, cognitive training exercises may delay the onset of more severe symptoms and improve the quality of life for those with Alzheimer’s disease. Although the extent to which such training can alter the disease’s trajectory is still under research, it is considered a beneficial component of a multi-faceted approach to Alzheimer’s care.
Social Engagement
Social engagement is another pillar of Alzheimer’s disease management, with growing evidence suggesting it may slow cognitive decline. Human interaction and socialization can stimulate brain activity, helping to maintain cognitive functioning and potentially delay the progression of Alzheimer’s symptoms. Social activities encourage the use of language, memory, and executive function skills, which are often affected in Alzheimer’s disease. Engaging in conversations, participating in group activities, and being part of a community can provide emotional support, reduce feelings of isolation, and promote a sense of belonging and purpose. Moreover, social engagement is often associated with better physical health and reduced stress, factors that contribute to an overall healthy lifestyle that supports brain health. While research is still uncovering the full extent of the benefits, maintaining strong social connections is commonly advised for those diagnosed with Alzheimer’s as part of a comprehensive approach to care that emphasizes the importance of mental and emotional well-being in disease progression.
Managing Comorbid Conditions
Effective management of cardiovascular risk factors such as hypertension, diabetes, obesity, and hypercholesterolemia can potentially influence the progression of Alzheimer’s disease. Ensuring that patients receive appropriate treatment for these conditions is a critical component of their overall care plan.
Experimental Approaches and Research
Researchers are continuously exploring new treatments to slow or halt the progression of Alzheimer’s disease. These include:
- Anti-Amyloid Treatments: Drugs aimed at reducing amyloid levels in the brain are a significant focus of Alzheimer’s research, with some showing promise in early clinical trials.
- Tau-Targeted Therapies: As tau tangles are another hallmark of Alzheimer’s, targeting them with specific therapies is an active area of investigation.
- Neuroprotective Agents: Compounds that can protect neurons from the toxic effects of amyloid and tau or counteract neuroinflammation are also under study.
Hope on the Horizon
Researchers at Case Western Reserve University have discovered a new approach to slow Alzheimer’s disease progression by inhibiting Slingshot Homolog-1 (SSH1), a protein that hinders the brain’s defense against oxidative stress. Oxidative stress leads to the brain’s corrosion and is a key initiator of Alzheimer’s and dementia. The study, led by David E. Kang and published in PNAS, demonstrated that deactivating SSH1 could increase the activation of Nuclear factor erythroid 2-related factor 2 (Nrf2), reducing oxidative damage and the accumulation of harmful brain plaques and tangles. This breakthrough suggests that enhancing the Nrf2 pathway could maintain neuron connections and prevent degeneration, a notable shift from the focus of current clinical trials, which largely address symptom management in advanced stages of Alzheimer’s. The recent FDA approval of Leqembi for early Alzheimer’s treatment and the development of SSH1 inhibitors underscore a promising direction for future therapeutic strategies aimed at the early stages of Alzheimer’s to potentially improve outcomes.
Conclusion
While there’s no definitive method for stopping Alzheimer’s disease once it starts, a combination of medication, lifestyle changes, and proactive management of comorbid conditions can help manage symptoms and potentially slow the rate of decline. Engaging in a healthy, active, and socially integrated lifestyle, combined with regular medical care, remains the best current approach to living with Alzheimer’s disease.
The quest to understand and fight Alzheimer’s disease is ongoing, and with each new study and clinical trial, we edge closer to more effective ways to control, cure, or prevent this condition. The partnership between patients, healthcare providers, and researchers is crucial as we continue to seek out hopeful interventions for this challenging disease.

Dr. Kashouty, a diplomate of the American Board of Psychiatry and Neurology (ABPN), practices general neurology with fellowship trained specialization in clinical neurophysiology. Dr. Kashouty finds the form and function of the nerves and muscles the most interesting part of neurology, which is what led him to specialize in neurophysiology with more emphasis on neuromuscular conditions. He treats all neurological diseases, but his main focus is to treat and manage headaches, movement disorders and neuromuscular diseases.





